Isotonic, hypotonic, and hypertonic solutions are three categories of IV fluids classified by their solute concentration relative to blood plasma. Each type triggers a different osmotic response at the cellular level, making fluid selection one of the most consequential decisions in clinical care.
This guide covers tonicity fundamentals and osmosis, isotonic fluid indications, hypotonic fluid indications, hypertonic fluid indications, key differences between all three solution types, nursing safety considerations, and how IV fluid principles apply to everyday hydration and recovery.
Tonicity defines whether cells maintain equilibrium, swell with incoming water, or shrink as water exits. Osmosis is the mechanism driving these shifts, and clinicians match a solution’s osmolarity to the patient’s specific deficit to determine which fluid to administer.
Isotonic solutions like 0.9% sodium chloride and Lactated Ringer’s match plasma osmotic pressure, producing no net fluid shift. They serve as first-line choices for volume expansion during hemorrhage, shock, severe vomiting, and diarrhea. Lactated Ringer’s offers a balanced electrolyte profile that may reduce adverse kidney events compared to standard saline.
Hypotonic solutions such as 0.45% and 0.225% sodium chloride lower vascular osmolality, driving water into dehydrated cells. They treat hypernatremia and cellular dehydration but are strictly contraindicated in patients with increased intracranial pressure due to the risk of cerebral edema.
Hypertonic solutions including 3% saline, D10W, D5NS, and D5LR pull water out of cells into the extracellular space. They address severe hyponatremia, cerebral edema, and hypoglycemia, though administration demands ICU-level monitoring to prevent osmotic demyelination syndrome.
Safe administration requires continuous tracking of vital signs, urine output, serum electrolytes, lung sounds, and neurological status. Selecting the wrong tonicity can cause fluid overload, pulmonary edema, or irreversible neurological damage.
What Are Isotonic, Hypotonic, and Hypertonic Solutions?
Isotonic, hypotonic, and hypertonic solutions are three categories of IV fluids classified by their solute concentration relative to blood plasma. The following sections explain tonicity and the role osmosis plays in fluid selection.
What Does Tonicity Mean in IV Fluid Therapy?
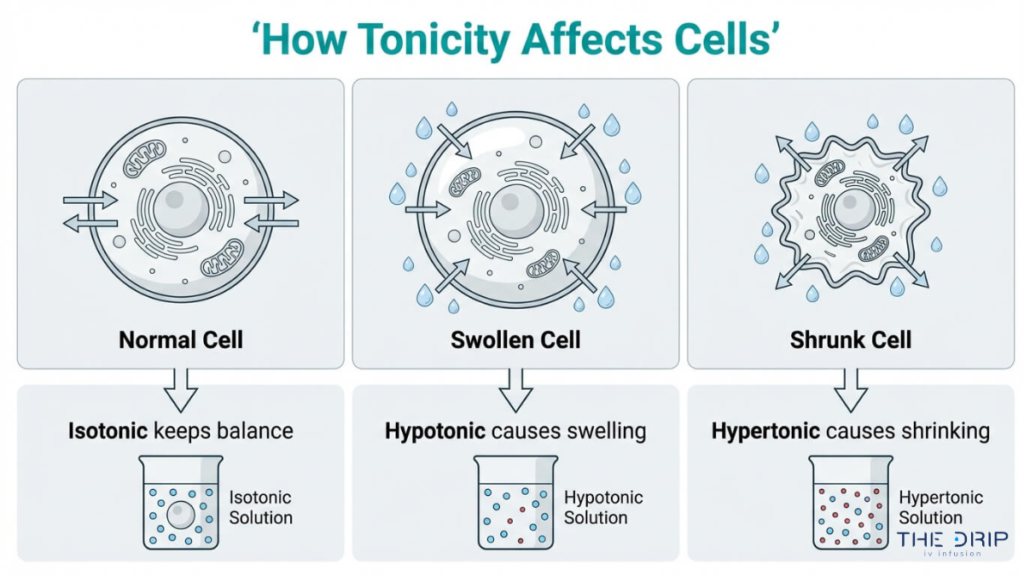
Tonicity in IV fluid therapy means the ability of an intravenous solution to influence water movement across cell membranes based on its solute concentration compared to plasma. This property determines whether cells maintain their shape, swell with excess water, or shrink as water is drawn out.
Clinicians rely on tonicity to guide every fluid order because selecting the wrong concentration carries serious consequences. According to the Society of Critical Care Medicine, complications of incorrect tonicity administration include fluid overload, pulmonary edema, cerebral edema, osmotic demyelination syndrome, and hyperchloremic metabolic acidosis.
To catch problems early, nurses monitor several parameters during infusion:
- Vital signs and hemodynamic trends
- Urine output volume and concentration
- Serum electrolyte levels
- Lung sounds, specifically crackles that signal fluid overload
- Neurological status changes
Understanding tonicity before choosing a solution is foundational; without it, even a routine fluid order can become dangerous.
How Does Osmosis Relate to IV Solution Selection?
Osmosis relates to IV solution selection because it is the mechanism through which IV fluids alter cellular hydration. When a solution enters the bloodstream, water moves across cell membranes from areas of lower solute concentration toward areas of higher solute concentration until equilibrium is reached.
According to Khan Academy, tonicity refers to the ability of an extracellular solution to make water move into or out of a cell by osmosis, which is directly related to the solution’s osmolarity. An isotonic fluid keeps water balanced across the membrane. A hypotonic fluid drives water into cells, while a hypertonic fluid pulls water out.
Clinicians match a solution’s osmolarity to the patient’s specific fluid deficit, making osmosis the governing principle behind every IV fluid decision. With these foundational concepts established, the next sections examine how each solution type behaves at the cellular level.
What Is an Isotonic Solution and How Does It Work?
An isotonic solution matches the osmotic pressure of blood plasma, preventing net fluid movement into or out of cells. The sections below explain cellular behavior in isotonic environments and the most common isotonic IV fluids used clinically.
What Happens to Cells in an Isotonic Solution?
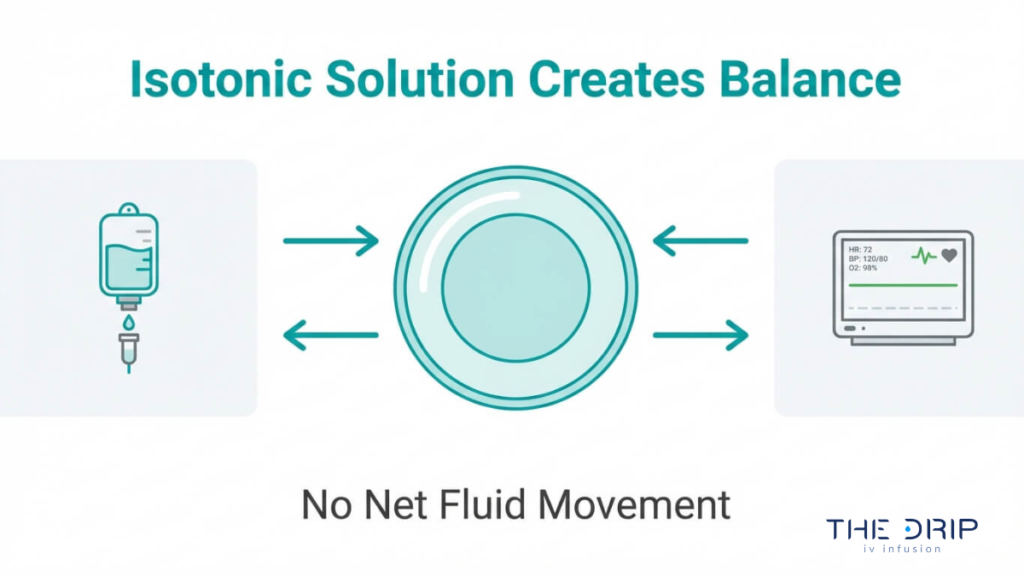
Cells in an isotonic solution remain in a state of equilibrium, with no net water movement across cell membranes. Because the solute concentration outside the cell equals the concentration inside, osmotic pressure is balanced in both directions.
According to Osmosis by Elsevier, isotonic solutions such as 0.9% Sodium Chloride (Normal Saline) and Lactated Ringer’s have the same osmotic pressure as plasma, causing no net fluid shift and keeping cells stable.
This equilibrium is precisely why isotonic fluids are preferred when the clinical goal is volume replacement without altering cell size. Neither swelling nor shrinkage occurs, which protects red blood cells and tissue integrity during rapid infusion.
What Are Common Examples of Isotonic IV Fluids?
Common examples of isotonic IV fluids include:
- 0.9% Sodium Chloride (Normal Saline): The most widely used crystalloid for general fluid resuscitation.
- Lactated Ringer’s Solution: A balanced electrolyte solution that closely mirrors plasma composition.
- 5% Dextrose in Water (D5W): Initially isotonic in the bag, though it becomes hypotonic once dextrose is metabolized.
According to the National Center for Biotechnology Information, isotonic fluids are the primary choice for volume expansion in clinical scenarios including hemorrhage, severe vomiting, diarrhea, shock, and metabolic acidosis. For clinicians and patients alike, understanding which isotonic fluid fits a given situation is foundational before considering hypotonic or hypertonic alternatives.
When Should You Give an Isotonic Solution?
You should give an isotonic solution when a patient needs intravascular volume expansion without shifting fluid into or out of cells. The specific isotonic fluid depends on the clinical scenario, electrolyte needs, and patient history.
When Should You Give Normal Saline (0.9% NaCl)?
You should give normal saline (0.9% NaCl) when a patient requires rapid volume resuscitation or has significant sodium and chloride losses. Common indications include:
- Hemorrhage and hypovolemic shock requiring immediate fluid replacement
- Severe dehydration from vomiting or diarrhea
- Blood transfusion compatibility, since normal saline is the only IV fluid safe to administer alongside blood products
- Diabetic ketoacidosis as the initial resuscitation fluid
- Metabolic alkalosis, where the chloride content helps correct the acid-base imbalance
Normal saline remains the most widely used crystalloid in emergency settings. However, prolonged high-volume infusion can contribute to hyperchloremic metabolic acidosis, making balanced alternatives worth considering for extended resuscitation.
When Should You Give a Lactated Ringer’s Solution?
You should give Lactated Ringer’s solution when replacing water and electrolyte loss in patients with low blood volume or low blood pressure. According to Mayo Clinic, Lactated Ringer’s is specifically indicated for these conditions but is contraindicated in patients with severe renal insufficiency due to the risk of fluid or solute overload.
Because Lactated Ringer’s contains potassium, calcium, and lactate in concentrations that closely mirror plasma, it causes less acid-base disruption than normal saline during large-volume resuscitation. Clinical research reported by the New England Journal of Medicine indicates that replacing standard saline with balanced crystalloids like Lactated Ringer’s in intensive care units can significantly reduce major adverse kidney events and death. For surgical patients and trauma cases, this balanced composition makes Lactated Ringer’s a preferred first-line isotonic fluid.
When Should You Give D5W as an Isotonic Infusion?
You should give D5W as an isotonic infusion when a patient needs a vehicle for intravenous medication delivery or requires modest caloric supplementation without additional sodium. D5W is isotonic in the bag, which means it does not cause vein irritation during administration.
Once infused, however, the body rapidly metabolizes the dextrose, leaving behind free water that distributes across all fluid compartments. This makes D5W unsuitable for true volume expansion. Clinicians typically select D5W for patients who need a keep-vein-open solution, those on sodium-restricted diets, or individuals requiring short-term hydration where electrolyte supplementation is unnecessary. Understanding this dual behavior is essential when selecting between isotonic fluids that restore volume and those that primarily deliver free water.
What Is a Hypotonic Solution and How Does It Work?
A hypotonic solution is an IV fluid with lower osmolality than blood plasma, causing water to move into cells through osmosis. The sections below explain how cells respond to hypotonic environments and which IV fluids fall into this category.
What Happens to Cells in a Hypotonic Solution?
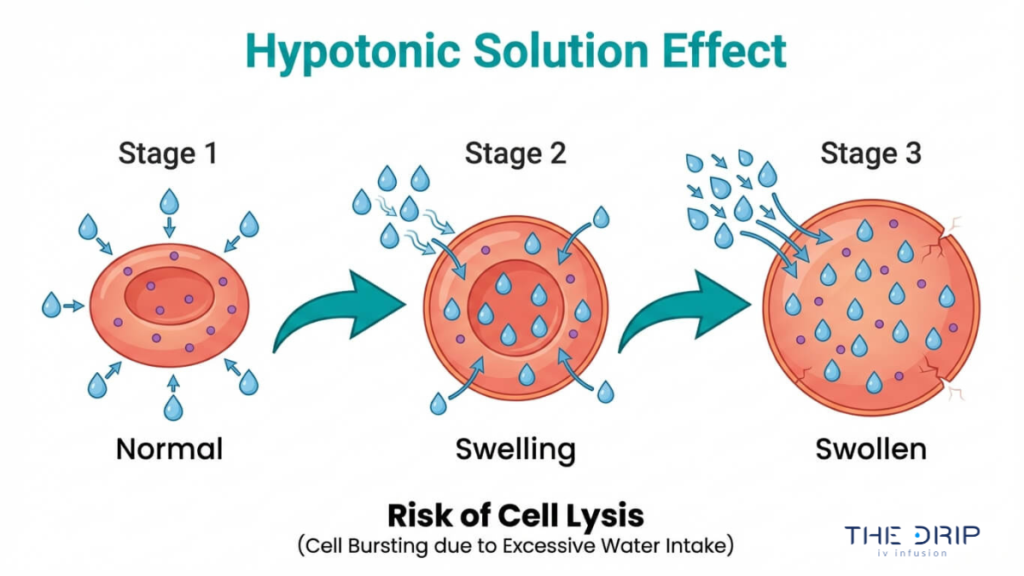
Cells in a hypotonic solution absorb water through osmosis because the surrounding fluid contains fewer dissolved particles than the intracellular environment. This inward fluid shift causes cells to swell, and in extreme cases, the membrane can rupture in a process called lysis.
According to CIA Medical, hypotonic solutions such as 0.45% Sodium Chloride (Half-Normal Saline) lower the osmolality within the vascular space, causing fluid to shift into the cells and potentially leading to cellular swelling or lysis. This mechanism makes hypotonic fluids useful for treating cellular dehydration and hypernatremia, where cells have lost water and need rehydration at the intracellular level.
However, this same fluid-shifting property creates serious risks in certain patients. Hypotonic solutions are strictly contraindicated in patients with increased intracranial pressure because the resulting cellular swelling can exacerbate cerebral edema. For clinicians, understanding this balance between therapeutic benefit and potential harm is what separates safe hypotonic fluid use from dangerous administration.
What Are Common Examples of Hypotonic IV Fluids?
The common examples of hypotonic IV fluids include solutions with osmolality at or below 250 mOsm/L. The most frequently used options are:
- 0.45% Sodium Chloride (Half-Normal Saline): The most widely administered hypotonic IV fluid, used for cellular rehydration and gradual sodium correction.
- 0.225% Sodium Chloride (Quarter-Normal Saline): An extremely hypotonic solution reserved for critical care settings when significant free water delivery is needed.
- D5W (post-metabolism): Initially isotonic in the bag, 5% Dextrose in Water becomes functionally hypotonic once the body metabolizes the glucose, leaving free water that distributes across fluid compartments.
Each fluid serves a distinct clinical purpose based on how aggressively intracellular rehydration needs to occur. With these foundational hypotonic fluids defined, the next step is understanding when each one should be administered.
When Should You Give a Hypotonic Solution?
You should give a hypotonic solution when a patient presents with cellular dehydration or elevated sodium levels. The specific clinical scenario determines whether 0.45% saline, 0.225% saline, or D5W is most appropriate.
When Should You Use 0.45% Normal Saline?
You should use 0.45% normal saline when treating hypernatremia and intracellular dehydration. This half-normal saline solution lowers vascular osmolality, driving water into dehydrated cells to restore fluid balance. Common indications include:
- Diabetic ketoacidosis maintenance fluids after initial isotonic resuscitation
- Hypernatremia requiring gradual sodium correction
- Daily maintenance fluid therapy for patients who cannot take fluids orally
According to Drugs.com, hypotonic solutions like 0.45% sodium chloride are strictly contraindicated in patients with increased intracranial pressure because they can exacerbate cerebral edema. Careful serum sodium monitoring during infusion prevents overly rapid correction, which risks neurological complications. For most clinical teams, this remains the default hypotonic choice when moderate free water delivery is needed.
When Should You Use 0.225% Normal Saline?
You should use 0.225% normal saline in specific critical care settings where aggressive free water replacement is necessary. This quarter-normal saline is an extremely hypotonic solution, making it far more potent at shifting fluid into cells than half-normal saline.
Its primary indication is severe hypernatremia requiring controlled free water administration. According to a study published in PubMed (National Library of Medicine), 0.225% sodium chloride is used in critical care to treat severe hypernatremia when free water delivery is required. Because of its very low osmolality, this solution carries a higher risk of rapid cellular swelling. Administration typically requires ICU-level monitoring with frequent electrolyte checks.
When Should You Use D5W as a Hypotonic Infusion?
You should use D5W as a hypotonic infusion when a patient needs free water without additional sodium. D5W is initially isotonic in the bag, but once the body metabolizes the dextrose, the remaining solution becomes hypotonic. This provides free water to both extracellular and intracellular compartments.
Key clinical uses include:
- Providing free water for patients with hypernatremia who need sodium-free hydration
- Serving as a drug diluent for IV medication delivery
- Supporting mild caloric supplementation in patients unable to eat
Clinicians often underestimate how quickly D5W’s tonicity shifts after infusion, which makes ongoing monitoring essential. Understanding these hypotonic options helps clinicians select the right concentration for each clinical scenario, a principle that applies equally when choosing hypertonic solutions.
What Is a Hypertonic Solution and How Does It Work?
A hypertonic solution is an IV fluid with higher solute concentration than blood plasma, which pulls water out of cells through osmosis. The sections below explain cellular response and common hypertonic fluid examples.
What Happens to Cells in a Hypertonic Solution?
Cells in a hypertonic solution lose water and shrink through a process called crenation. Because the surrounding fluid contains more dissolved particles than the cell’s interior, osmosis drives water outward across the cell membrane to equalize concentration. This deliberate fluid shift is precisely why clinicians select hypertonic solutions: to reduce dangerous swelling in tissues, particularly the brain.
According to RxList, hypertonic solutions such as 3% Sodium Chloride create an osmotic pressure gradient that draws water out of the intracellular space into the extracellular space, causing cells to shrink. Given this potent effect, administration requires careful oversight. State nursing boards regulate who can manage these infusions; Mississippi, for example, specifically prohibits LPNs from independently regulating dosages of hypertonic solutions. For most clinical scenarios, hypertonic fluids demand central venous access and continuous monitoring because rapid osmotic shifts can cause serious complications if poorly controlled.
What Are Common Examples of Hypertonic IV Fluids?
Common examples of hypertonic IV fluids include 3% Sodium Chloride, 5% Sodium Chloride, D10W, D5NS, and D5LR. Each serves a distinct clinical purpose:
- 3% Sodium Chloride (3% NaCl) is used for severe hyponatremia and cerebral edema, requiring slow, monitored infusion.
- 5% Sodium Chloride (5% NaCl) treats critically low sodium levels under intensive care supervision.
- D10W (10% Dextrose in Water) provides 340 calories per liter and addresses hypoglycemia or nutritional support needs.
- D5NS (5% Dextrose in 0.9% NaCl) combines electrolyte replacement with caloric supplementation while maintaining high osmolarity.
- D5LR (5% Dextrose in Lactated Ringer’s) delivers electrolytes and calories, preventing fluid from shifting into cells.
The variety within this category highlights an important point: not all hypertonic fluids work the same way. Selecting the right one depends on whether the clinical goal is sodium correction, glucose support, or combined electrolyte and caloric delivery. Understanding when each is appropriate is essential for safe IV fluid management.
When Should You Give a Hypertonic Solution?
You should give a hypertonic solution when a patient needs rapid fluid shift from cells into the vascular space. Common clinical scenarios include severe hyponatremia, cerebral edema, and hypoglycemia requiring concentrated glucose.
When Should You Use 3% Saline?
You should use 3% saline when a patient presents with severe, symptomatic hyponatremia or acute cerebral edema. This concentrated sodium chloride solution creates a strong osmotic gradient that pulls excess water out of swollen brain cells, reducing dangerous intracranial pressure.
According to the U.S. Food and Drug Administration, 3% and 5% sodium chloride injections are indicated for treating severe hyponatremia and cerebral edema, though they must be administered with extreme caution to avoid osmotic demyelination syndrome. Because of this risk, 3% saline typically requires ICU-level monitoring with frequent serum sodium checks. For clinicians, the critical principle is controlled correction; raising sodium too quickly can cause irreversible neurological damage, making this one of the highest-risk IV fluids in practice.
When Should You Use D10W?
You should use D10W when a patient requires treatment for hypoglycemia or needs concentrated caloric supplementation through an IV line. D10W, or 10% dextrose in water, is a hypertonic solution that delivers a higher glucose load than standard dextrose fluids.
According to DailyMed, D10W provides 340 calories per liter and is used for nutritional support or to treat hypoglycemia when higher glucose concentrations are required. This makes it particularly valuable for neonates, patients who cannot eat, or those with persistently low blood sugar unresponsive to D5W. Blood glucose levels should be monitored closely during infusion, since overcorrection can lead to hyperglycemia and osmotic complications.
When Should You Use D5 in 0.9% Normal Saline?
You should use D5 in 0.9% normal saline (D5NS) when a patient needs both fluid volume expansion and supplemental calories. D5NS combines the isotonic electrolyte profile of normal saline with 5% dextrose, creating a hypertonic solution that maintains high osmolarity while delivering energy.
Common indications include postoperative fluid maintenance, patients with combined caloric and electrolyte deficits, and situations where prolonged NPO (nothing by mouth) status demands nutritional support alongside hydration. Because D5NS provides sodium, chloride, and glucose simultaneously, it is well suited for patients who need more than simple rehydration. Serum glucose and electrolytes should be monitored throughout the infusion.
When Should You Use D5 in Lactated Ringers?
You should use D5 in Lactated Ringer’s (D5LR) when a patient requires balanced electrolyte replacement combined with caloric intake. D5LR pairs the multi-electrolyte composition of Lactated Ringer’s, including sodium, potassium, calcium, and lactate, with 5% dextrose for energy.
This solution is frequently selected for surgical patients, burn recovery, and prolonged fluid therapy where both electrolyte balance and caloric support matter. Compared to D5NS, D5LR offers a more physiologically balanced electrolyte profile that reduces the risk of hyperchloremic metabolic acidosis. However, it remains contraindicated in patients with severe hepatic impairment, since the liver must metabolize lactate to bicarbonate. Selecting between D5NS and D5LR ultimately depends on the patient’s acid-base status and electrolyte needs.
What Are the Key Differences Between Isotonic, Hypotonic, and Hypertonic Solutions?
The key differences between isotonic, hypotonic, and hypertonic solutions center on osmolality, the direction of water movement across cell membranes, and their clinical applications. Normal serum osmolality falls between 275 and 295 mOsm/kg, according to StatPearls, and each solution type is defined by its concentration relative to this baseline.
| Feature | Isotonic | Hypotonic | Hypertonic |
| Osmolality Range | 250–375 mOsm/L | ≤ 250 mOsm/L | ≥ 375 mOsm/L |
| Water Movement | No net fluid shift | Water moves into cells | Water moves out of cells |
| Cell Response | Equilibrium | Swelling (possible lysis) | Shrinkage (crenation) |
| Primary Use | Volume expansion, shock, hemorrhage | Cellular dehydration, hypernatremia | Severe hyponatremia, cerebral edema |
| Common Examples | 0.9% NaCl, Lactated Ringer’s | 0.45% NaCl, 0.225% NaCl | 3% NaCl, D10W, D5NS |
Understanding these distinctions matters because selecting the wrong tonicity can worsen a patient’s condition rather than correct it. For clinicians and nursing students frequently searching for clarification on isotonic versus hypotonic solutions for dehydration, this comparison serves as a practical reference point. With these differences established, proper monitoring during administration becomes the next critical step.
What Nursing Considerations Apply When Administering IV Solutions?
Nursing considerations when administering IV solutions include continuous patient monitoring, accurate tonicity selection, and awareness of complications from incorrect fluid choices. The subsections below cover monitoring protocols and risks of tonicity errors.
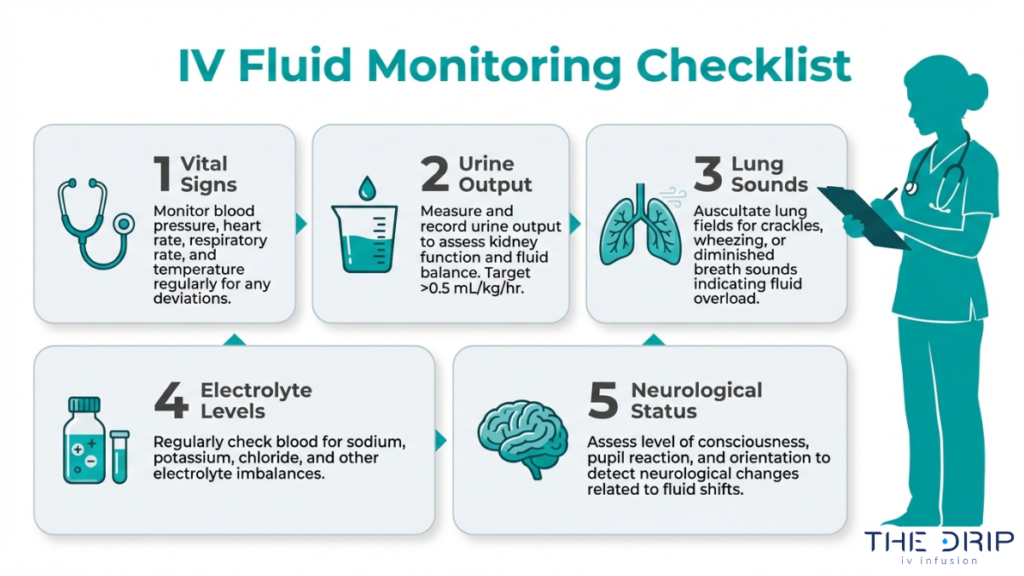
How Do You Monitor a Patient Receiving IV Fluids?
You monitor a patient receiving IV fluids by tracking a specific set of clinical parameters throughout the infusion. According to BC Campus Open Publishing, essential nursing monitoring parameters for IV therapy include vital signs, urine output, serum electrolytes, lung sounds for crackles indicating fluid overload, and neurological status.
Effective monitoring follows a consistent assessment cycle:
- Measure vital signs at baseline and at regular intervals during infusion.
- Track urine output to evaluate kidney function and fluid balance.
- Auscultate lung sounds for crackles, which signal early pulmonary edema.
- Review serum electrolyte levels to detect imbalances caused by the infusion.
- Assess neurological status, especially when administering hypotonic or hypertonic solutions.
Skipping any single parameter can mask early warning signs, making a structured checklist one of the most practical tools a nurse can use during IV administration.
What Are the Risks of Giving the Wrong IV Tonicity?
The risks of giving the wrong IV tonicity include fluid overload, pulmonary edema, cerebral edema, osmotic demyelination syndrome, and hyperchloremic metabolic acidosis. Each complication stems from unintended fluid shifts between the intravascular and intracellular compartments.
Administering a hypotonic solution to a patient with elevated intracranial pressure, for example, can worsen cerebral edema by driving water into brain cells. Conversely, correcting hyponatremia too rapidly with hypertonic saline risks osmotic demyelination syndrome, which causes irreversible neurological damage. Even isotonic solutions carry risk when infused in excessive volumes, potentially triggering pulmonary edema in patients with compromised cardiac function.
These consequences reinforce why tonicity selection requires careful assessment before and throughout every infusion. Understanding the relationship between solution type and complication risk forms the foundation for safe IV fluid delivery in any clinical or wellness setting.
How Does IV Fluid Tonicity Apply to Everyday Hydration Needs?
IV fluid tonicity applies to everyday hydration needs by informing how professionals select solutions that restore fluid balance outside hospital settings. The sections below cover mobile IV therapy for wellness recovery and key takeaways on isotonic, hypotonic, and hypertonic solution selection.
Can Mobile IV Therapy From The Drip IV Infusion Help With Rehydration and Recovery?
Yes, mobile IV therapy from The Drip IV Infusion can help with rehydration and recovery by delivering fluids, electrolytes, and vitamins directly into the bloodstream. This approach bypasses the GI tract, allowing maximum absorption for individuals experiencing dehydration from illness, athletic exertion, or general fatigue. The Drip IV Infusion provides customizable IV cocktails administered by experienced nurses in the comfort of a client’s home or workplace. Although a 2024 review published in the JAMA Network noted limited high-quality evidence supporting vitamin drips in healthy individuals, medically supervised IV rehydration remains a well-established method for replenishing fluids when oral intake is insufficient. For anyone seeking professional hydration support, The Drip IV Infusion offers convenient, nurse-administered sessions tailored to individual wellness goals.
What Are the Key Takeaways About When to Give Isotonic, Hypotonic, and Hypertonic Solutions?
The key takeaways about when to give isotonic, hypotonic, and hypertonic solutions center on matching a fluid’s osmolality to the patient’s clinical need:
- Isotonic solutions (250 to 375 mOsm/L) maintain plasma equilibrium and serve as first-line choices for volume replacement during hemorrhage, shock, or severe dehydration.
- Hypotonic solutions (≤250 mOsm/L) push water into cells, making them appropriate for cellular dehydration and hypernatremia while being contraindicated with elevated intracranial pressure.
- Hypertonic solutions (≥375 mOsm/L) pull water out of cells, treating conditions such as severe hyponatremia and cerebral edema under close monitoring.
One notable exception is D5W, which is initially isotonic in the bag but becomes hypotonic once dextrose is metabolized, providing free water to both extracellular and intracellular compartments. Understanding these classifications ensures each fluid choice aligns with the body’s immediate physiological demand.




