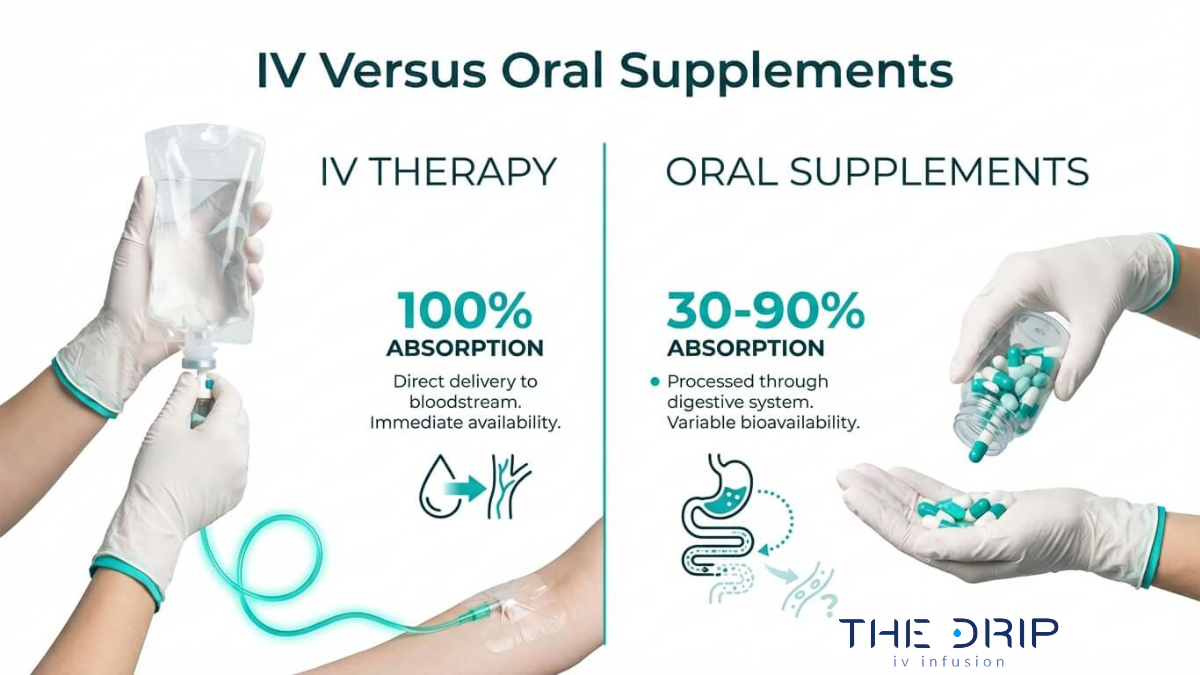
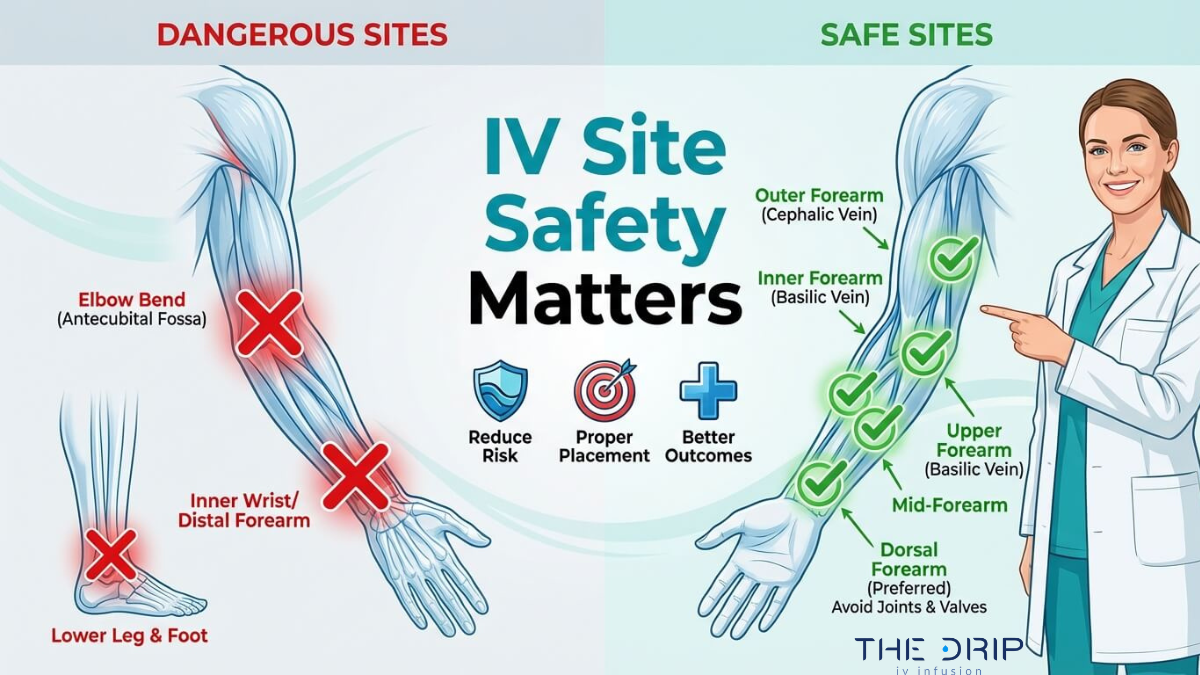
An IV hydration business in Arizona is a healthcare venture that requires a specific combination of legal formation, medical oversight, clinical licensing, and regulatory compliance before treating a single client. This guide covers business registration and legal structure, medical director and nursing requirements, clinical safety and supply chain compliance, startup and operational costs, and marketing regulations.
Arizona LLC formation starts at $50 through the Arizona Corporation Commission, with additional state and local tax licenses required before opening. Each municipality sets its own permit rules, so operators must verify local requirements city by city.
Medical director oversight is non-negotiable. A qualified physician with an active, unrestricted MD or DO license must approve all treatment protocols, issue individualized medical orders, and provide on-site supervision. Monthly fees for this oversight typically fall between $1,000 and $3,000 depending on clinic complexity and patient volume.
Every IV treatment must be administered by a licensed Registered Nurse or Licensed Practical Nurse operating under a patient-specific medical order. Arizona’s Board of Nursing has clarified that blanket standing orders for elective IV therapies do not satisfy the provider’s legal duty to each patient, making individualized assessments mandatory.
Clinical operations must meet OSHA bloodborne pathogen standards, USP compounding rules for sterile preparations, and Arizona biomedical waste disposal regulations. Sourcing IV fluids exclusively from FDA-registered manufacturers or 503B outsourcing facilities keeps the supply chain legally sound.
Total startup costs range from $6,000 for a mobile setup to over $40,000 for a stationary clinic, with ongoing expenses including insurance, supplies, and medical director fees. All advertising must comply with FTC truthfulness standards and HIPAA patient privacy rules.
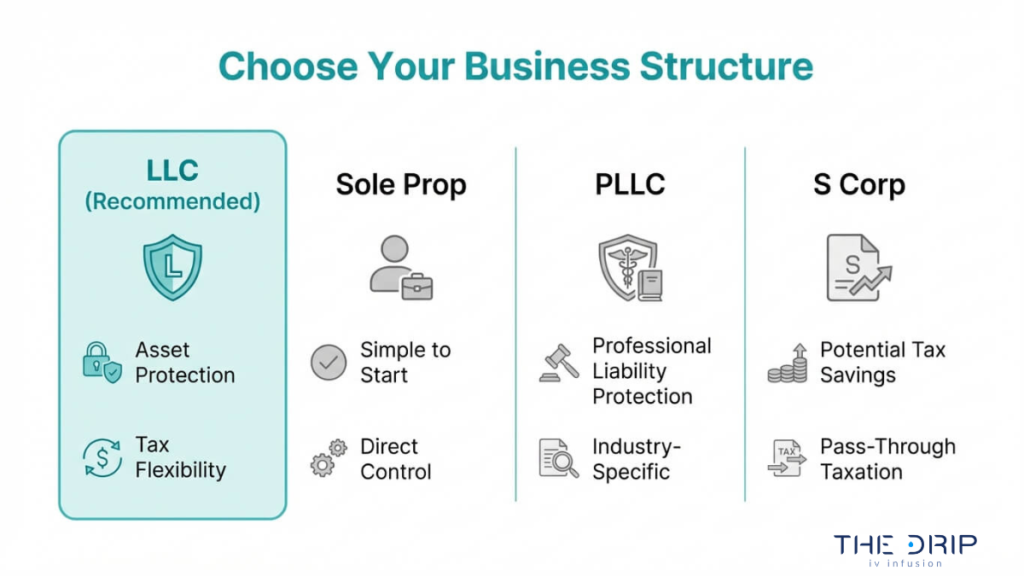
What Legal Structure Should You Choose for an IV Hydration Business?
The legal structure you should choose for an IV hydration business is typically a Limited Liability Company (LLC), which offers personal asset protection and operational flexibility suited to healthcare ventures. Below, this section covers why the LLC is the preferred entity type, how to file in Arizona, and what other structures to consider.
An LLC separates personal assets from business liabilities, a critical distinction when offering medical services like IV therapy. If a patient files a claim related to treatment, the LLC structure shields the owner’s personal finances from business debts and legal judgments. This protection layer works alongside professional liability insurance to create a more defensible position.
Arizona makes LLC formation straightforward and affordable. According to the Arizona Corporation Commission, the filing fee for Articles of Organization for a domestic LLC is $50 for regular processing and $85 for expedited processing. After filing, you will need to publish your Articles of Organization in an approved newspaper for three consecutive weeks, a requirement unique to Arizona.
While the LLC remains the most common choice, other structures exist:
- Sole proprietorship offers simplicity but provides zero liability protection, making it a poor fit for healthcare businesses.
- Professional LLC (PLLC) may apply if a licensed physician directly owns and operates the clinic.
- S-Corporation election allows LLC owners to reduce self-employment taxes once revenue reaches a sustainable level.
- Partnership suits multi-owner ventures but still requires an LLC wrapper for liability coverage.
For most IV hydration entrepreneurs, the standard LLC paired with an S-Corp tax election once profitable strikes the right balance between protection and tax efficiency. Owners who are not licensed healthcare providers can still form the LLC, since Arizona allows non-physicians to own IV hydration businesses as long as licensed professionals deliver all clinical care under proper medical oversight.
Beyond formation, the LLC will need an Employer Identification Number (EIN) from the IRS, which is free and available online. This number is required to open a business bank account, hire staff, and file taxes. Keeping business and personal finances completely separate from day one strengthens the liability shield the LLC provides.
Choosing the right structure early prevents costly restructuring later, especially once licensing and insurance requirements build on top of your entity type.
What Arizona Business Licenses and Registrations Are Required?
Arizona business licenses and registrations required for an IV hydration business include a Transaction Privilege Tax license, Arizona Corporation Commission registration, and local city or county permits. Each layer of compliance is covered below.
What Is the Arizona Transaction Privilege Tax License?
The Arizona Transaction Privilege Tax (TPT) license is a state-issued authorization that allows businesses to conduct taxable activities. Any IV hydration business collecting payment for services must obtain this license before operating.
According to the Arizona Department of Revenue, the TPT license fee is $12 per business location and must be renewed annually by January 1st. Local municipalities often add their own fees on top. In Scottsdale, for example, the initial city-level TPT fee is $50, charged in addition to the state fee.
Applying early matters. Because renewal deadlines are fixed, securing the license well before launch prevents gaps in compliance that could delay opening day.
How Do You Register with the Arizona Corporation Commission?
You register with the Arizona Corporation Commission (ACC) by filing formation documents that match your chosen business structure. An LLC, for instance, requires Articles of Organization submitted directly to the ACC.
The standard filing fee for a domestic LLC is $50, while expedited processing costs $85. For faster turnaround, same-day service is available at $200, provided documents arrive by 10:00 a.m. Registration through the ACC establishes the business as a legal entity in Arizona, which is a prerequisite for obtaining tax licenses, opening business bank accounts, and entering contracts. Keeping the entity in good standing requires timely annual report filings with the Commission.
What Local City or County Permits Are Needed?
Local city or county permits needed for an IV hydration business vary significantly by municipality. Arizona has no uniform local licensing framework, so requirements depend entirely on where the business operates.
- Phoenix does not issue a general business license; instead, it regulates specific activities through individual regulatory permits.
- Scottsdale requires a separate city-level TPT license and may impose additional zoning or use permits.
- Gilbert, Mesa, and other cities each maintain their own permit structures for health-related services.
Contacting the local planning and zoning department before signing a lease is the most reliable way to confirm which permits apply. Zoning restrictions can disqualify certain commercial spaces for healthcare use entirely, making early verification essential. With licensing and registration secured, the next step is establishing medical director oversight.
What Are the Medical Director Requirements for IV Therapy in Arizona?
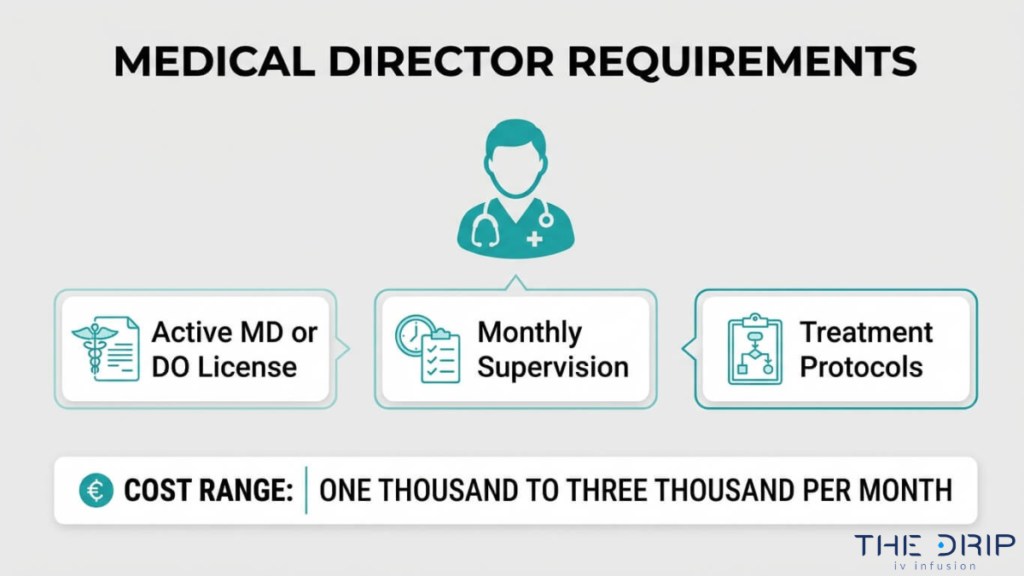
The medical director requirements for IV therapy in Arizona center on physician oversight, proper licensing, a formal agreement, and ongoing compliance. The following subsections cover the medical director’s role, required qualifications, agreement structure, and typical costs.
What Does a Medical Director Do for an IV Hydration Business?
A medical director for an IV hydration business serves as the physician responsible for all clinical protocols, medical orders, and patient safety oversight. According to Goldberg Law Office, “The Medical Director is the cornerstone of a compliant IV hydration business, ensuring that every treatment is backed by a valid medical order and that all clinical staff are operating within their legal scope of practice.”
In Arizona, anyone can own an IV hydration business, but licensed professionals must deliver care under physician oversight. Core medical director responsibilities include:
- Establishing and approving treatment protocols for each IV infusion offered.
- Issuing individualized medical orders that authorize nurses to administer therapy.
- Verifying that all clinical staff operate within their legal scope of practice.
- Reviewing patient intake records and ensuring proper documentation.
- Overseeing adverse reaction procedures and emergency protocols.
This role is not ceremonial. Without active medical director involvement, an IV hydration business in Arizona risks operating outside legal compliance, which can result in board investigations or license revocations.
What Qualifications Must an Arizona Medical Director Have?
An Arizona medical director must hold an active, unrestricted MD or DO license from the Arizona Medical Board. Board certification in a relevant specialty, while not always mandated by statute, strengthens compliance credibility and demonstrates clinical competence in overseeing infusion services.
Beyond licensure, Arizona oversight rules require medical directors to provide direct, in-person, on-site supervision for at least four hours per month in infusion settings, according to Quarles & Brady LLP. Additional qualification requirements include:
- Familiarity with IV therapy protocols, including hydration, vitamin infusion, and NAD+ administration.
- Knowledge of Arizona recordkeeping laws, which mandate maintaining legible medical records for at least six years after the last date of service.
- Malpractice insurance coverage appropriate for supervisory responsibilities.
Selecting a medical director with hands-on infusion experience, rather than one who simply holds a license, significantly reduces compliance risk and improves clinical quality.
How Do You Structure a Medical Director Agreement?
A medical director agreement is a formal written contract that defines the scope of physician oversight, responsibilities, and compensation terms between the medical director and the IV hydration business. Every compliant agreement should address these core elements:
- Scope of services, specifying which protocols the director supervises and approves.
- On-site supervision schedule, including minimum monthly hours.
- Compensation structure, whether monthly retainer, hourly rate, or hybrid.
- Liability allocation and malpractice insurance requirements for both parties.
- Termination clauses with defined notice periods.
- Chart review frequency and quality assurance obligations.
The agreement should never create an employment relationship unless intended; most IV businesses structure this as an independent contractor arrangement. Having a healthcare attorney review the contract before execution protects both the business owner and the medical director from regulatory gaps.
How Much Does a Medical Director Cost in Arizona?
A medical director in Arizona typically costs between $1,000 and $3,000 per month, or $150 to $300 per hour, depending on the level of involvement and number of staff supervised. According to ZipRecruiter, these ranges reflect current market rates for IV hydration clinic oversight roles.
Several factors influence the final cost:
- Number of clinic locations or mobile service areas requiring oversight.
- Volume of patients and chart reviews performed monthly.
- Whether the director provides on-site supervision beyond the minimum four hours.
- Complexity of the treatment menu, since specialty infusions like NAD+ may require deeper clinical involvement.
Professional liability policies add another layer of cost; coverage for IV hydration businesses can include up to $25,000 per claim and $100,000 aggregate for medical expenses from covered incidents. For most new IV hydration businesses, budgeting $2,000 to $3,000 monthly for a responsive, experienced medical director is a practical starting point.
With medical director requirements clearly defined, the next step is understanding how Arizona Board of Nursing regulations shape the clinical team delivering IV therapy.
What Arizona Board of Nursing Regulations Apply to IV Therapy?
Arizona Board of Nursing regulations for IV therapy govern scope of practice, required medical orders, and supervision standards for nurses who administer infusions. The sections below cover RN scope, LPN permissions, and supervision requirements.
What Scope of Practice Do Registered Nurses Have for IV Infusion?
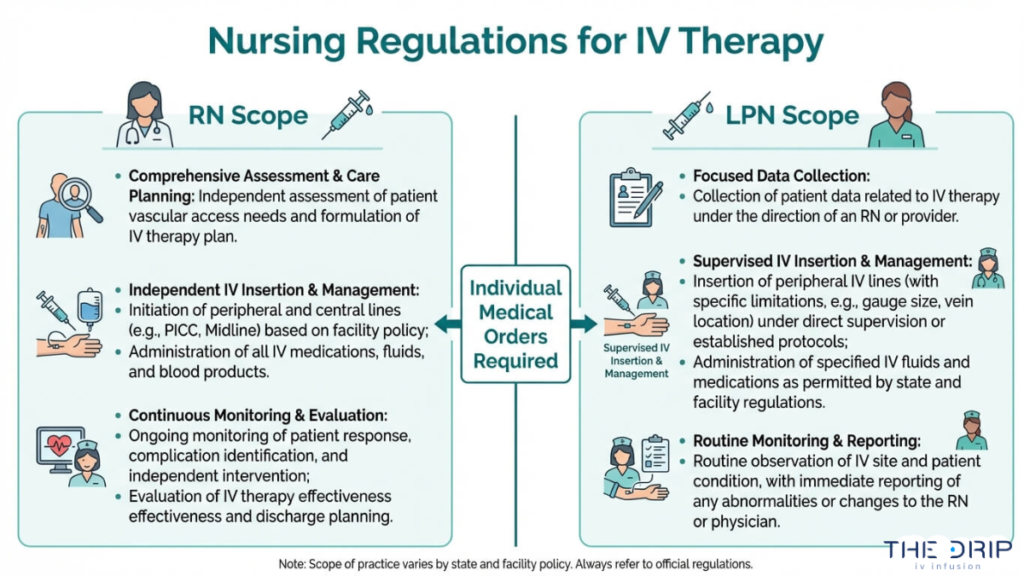
The scope of practice for Registered Nurses performing IV infusion in Arizona includes patient assessment, venipuncture, IV catheter insertion, infusion monitoring, and adverse reaction management. RNs operate under the authority of the Arizona State Board of Nursing and must follow individualized medical orders issued by a qualified Licensed Provider.
Each order must reflect a prior patient examination and established provider-patient relationship. Standing orders alone do not satisfy this requirement for elective IV therapies. RNs may not independently prescribe IV treatments or modify protocols without provider authorization.
According to Quarles & Brady LLP, qualified practitioners must provide direct, in-person, on-site supervision for at least four hours per month in certain infusion settings to remain compliant with Arizona oversight rules.
Can Licensed Practical Nurses Administer IV Therapy in Arizona?
Yes, Licensed Practical Nurses can administer IV therapy in Arizona, but only under specific conditions. According to the Arizona State Board of Nursing, both RNs and LPNs must have an individualized medical order from a qualified Licensed Provider who has established a patient relationship and completed an examination prior to administering IV therapy.
LPNs face additional restrictions compared to RNs. Their scope typically limits them to performing IV tasks under the direct supervision of an RN or physician, rather than practicing independently. LPN IV competency must be documented and verified before any infusion administration begins. For business owners staffing an IV hydration clinic, understanding this distinction between RN and LPN authority is essential for maintaining compliance.
What Are the Supervision Requirements for IV Nurses in Arizona?
The supervision requirements for IV nurses in Arizona mandate that all nursing staff operate under the clinical oversight of a licensed physician or qualified provider. This oversight ensures every IV treatment traces back to an individualized medical order, not a blanket standing order.
Key supervision requirements include:
- A Licensed Provider must establish a patient relationship and complete an examination before issuing IV therapy orders.
- Standing orders issued by an APRN for other staff to follow are not consistent with Arizona State Board of Nursing advisory opinions.
- RNs may perform IV infusions under general supervision, while LPNs require direct supervision from an RN or physician.
- Clinical protocols, emergency procedures, and adverse event reporting must be documented and reviewed by the supervising provider.
Proper supervision structures protect both patients and the business from regulatory violations, making this one of the most critical compliance areas for any Arizona IV hydration operation.
What Standing Order and Protocol Requirements Must You Follow?
The standing order and protocol requirements you must follow in Arizona center on individualized patient assessments, valid medical orders from qualified providers, and proper documentation. Arizona takes a stricter position than many states on how IV therapy orders are issued.
Arizona’s regulatory framework distinguishes between individualized medical orders and blanket standing orders for elective IV therapies. The Arizona State Board of Nursing has issued advisory opinions clarifying that generic standing orders do not meet the provider’s legal duty to each patient. Instead, every IV hydration treatment requires a medical order tied to a specific patient evaluation.
This requirement means a qualified Licensed Provider, such as a physician (MD or DO) or nurse practitioner with prescriptive authority, must establish a patient relationship and complete an examination before authorizing IV therapy. Registered Nurses and Licensed Practical Nurses cannot independently initiate elective IV treatments based solely on pre-written protocols that lack individualized assessment.
According to the Arizona State Board of Nursing, the issuance of standing orders for elective IV therapies by an APRN for other healthcare staff to follow is not consistent with the Board’s advisory opinions and does not satisfy the provider’s duty to the patient.
Your IV hydration business protocols should address these key requirements:
- Each client must receive an individualized assessment before any infusion begins.
- A Licensed Provider must issue a patient-specific medical order based on that assessment.
- Clinical staff must verify the order before administering any IV treatment.
- All medical orders, assessments, and treatment records must be documented and maintained for at least six years, as required by Arizona law.
- Protocols must define the scope of each staff member’s role to ensure compliance with Arizona Board of Nursing regulations.
For businesses relying on telehealth consultations to fulfill the provider evaluation requirement, the same standards apply. The provider must still conduct a meaningful assessment and issue an individualized order rather than a blanket authorization.
Many IV hydration business owners underestimate the distinction between clinical standing orders used in hospital emergency settings and elective wellness protocols. In hospital settings, standing orders function under institutional oversight structures that do not exist in most IV hydration businesses. Applying that same model to an elective wellness clinic creates significant compliance risk.
Building compliant protocols from the start protects both the business and its clients. With protocols in place, securing appropriate insurance coverage becomes the next essential step.
What Insurance Do You Need for an IV Hydration Business in Arizona?
The insurance you need for an IV hydration business in Arizona includes professional liability, general liability, and workers compensation coverage. Each policy protects against different risks, from clinical errors to workplace injuries.
What Does Professional Liability Insurance Cover?
Professional liability insurance covers claims arising from errors, omissions, or negligence in the clinical services your IV hydration business provides. This includes allegations of improper IV administration, incorrect dosing, or adverse patient reactions linked to treatment decisions.
According to Insureon, professional liability insurance for small businesses in Arizona costs an average of $91 per month, though rates vary by industry and business size. For IV therapy operations specifically, policies may include coverage for medical expenses resulting from incidents during treatment. Given the hands-on clinical nature of IV infusion, this is arguably the most critical policy any hydration business should secure before treating a single client.
What Does General Liability Insurance Cover?
General liability insurance covers third-party bodily injury, property damage, and advertising injury claims unrelated to clinical treatment. If a client slips in your clinic lobby or you damage property at a mobile service location, this policy responds.
One often-overlooked element involves marketing compliance. HIPAA marketing rules generally prohibit covered entities from using or disclosing Protected Health Information for marketing purposes without valid written patient authorization, according to the U.S. Department of Health and Human Services. An advertising injury claim tied to improper use of patient data could fall under general liability. Pairing this coverage with strict HIPAA-compliant marketing practices reduces exposure significantly.
Is Workers Compensation Insurance Required in Arizona?
Yes, workers compensation insurance is required in Arizona for businesses that employ one or more workers. Arizona law mandates this coverage regardless of whether employees work full-time, part-time, or on a temporary basis.
Workers compensation pays for medical expenses, lost wages, and rehabilitation costs when an employee suffers a work-related injury or illness. For IV hydration businesses, common workplace risks include needlestick injuries, exposure to bloodborne pathogens, and repetitive strain from clinical procedures. Failing to carry this coverage can result in penalties, lawsuits, and personal liability for the business owner. Securing workers compensation before hiring your first infusion specialist is non-negotiable for compliant operations.
With insurance protections in place, understanding FDA and pharmacy regulations ensures your IV supply chain remains legally sound.
What Are the FDA and Pharmacy Regulations for IV Fluids in Arizona?
The FDA and pharmacy regulations for IV fluids in Arizona govern how clinics source injectable products, compound sterile preparations, and document their supply chain. The subsections below cover legal sourcing, compounding rules, and required supplier documentation.
How Do You Legally Source IV Fluids and Vitamins?
You legally source IV fluids and vitamins by purchasing them from FDA-registered manufacturers, licensed wholesalers, or 503B outsourcing facilities. The FDA classifies IV fluids and injectable vitamins as prescription drugs, which means every product entering your clinic must originate from a facility that meets Current Good Manufacturing Practice (CGMP) standards.
503B outsourcing facilities provide a practical sourcing path for IV hydration businesses. These facilities produce compounded sterile preparations in bulk without requiring patient-specific prescriptions, provided they remain registered with the FDA and submit to regular inspections. For operators wanting tighter compliance control, sourcing exclusively from 503B facilities rather than 503A compounding pharmacies reduces regulatory exposure considerably.
What Are Arizona State Board of Pharmacy Compounding Rules?
The Arizona State Board of Pharmacy compounding rules require IV hydration clinics that prepare sterile formulations to follow United States Pharmacopeia (USP) <797> standards for safe preparation. USP <797> establishes requirements for cleanroom environments, beyond-use dating, personnel training, and quality assurance testing.
According to the Alliance for Pharmacy Compounding, the Arizona State Board of Pharmacy moved to delay the enforcement of updated USP 795, 797, and 800 compounding standards as of September 2025. This delay gives clinics additional time to adapt, but it does not eliminate the underlying compliance obligation.
Clinics should also note that standing orders do not simplify compounding oversight. The Arizona State Board of Nursing has stated that issuing standing orders for elective IV therapies by an APRN is not consistent with its advisory opinions and does not satisfy the provider’s duty to the patient. Each compounded preparation still requires an individualized order tied to a specific patient assessment.
What Supplier Documentation Must You Maintain?
The supplier documentation you must maintain includes records that verify every IV fluid and injectable product traces back to a licensed, FDA-compliant source. Essential documents include:
- Certificates of Analysis (COAs) confirming purity, potency, and sterility for each product lot.
- Proof of supplier licensure, such as state pharmacy board registration or FDA 503B facility registration.
- Purchase invoices and packing slips showing lot numbers, quantities, expiration dates, and delivery dates.
- Beyond-use dating records for any compounded sterile preparations received from outsourcing facilities.
Maintaining organized supplier files protects your business during state board inspections and demonstrates regulatory diligence. Missing even one COA for a product lot can trigger compliance flags that are entirely preventable with a consistent filing system.
With sourcing and documentation protocols established, health and safety compliance standards address how these products are handled in your clinical environment.
What Health and Safety Compliance Standards Apply in Arizona?
Health and safety compliance standards that apply in Arizona include federal OSHA regulations, infection control protocols, biomedical waste disposal rules, and emergency preparedness requirements. The following subsections cover each area.
What Are the OSHA Requirements for IV Therapy Businesses?
The OSHA requirements for IV therapy businesses center on the Bloodborne Pathogens Standard (29 CFR 1910.1030), which applies to all occupational exposure to blood or other potentially infectious materials in healthcare settings, according to the Occupational Safety and Health Administration. IV hydration businesses must develop a written Exposure Control Plan that identifies at-risk tasks, mandates personal protective equipment such as gloves and face shields, and establishes procedures for handling sharps.
Staff training on bloodborne pathogen risks is required at the time of hire and annually thereafter. Employers must also provide hepatitis B vaccinations to employees with occupational exposure at no cost. Maintaining detailed training logs and incident records is essential for passing OSHA inspections.
What Infection Control Protocols Must You Implement?
The infection control protocols you must implement cover hand hygiene, aseptic technique, surface disinfection, and sharps safety. Every IV insertion requires a standardized aseptic technique using alcohol-based skin prep and sterile, single-use catheter supplies. Reusable equipment, including tourniquets and infusion chairs, must be disinfected between each client using EPA-registered hospital-grade disinfectants.
Hand hygiene compliance forms the foundation of infection prevention. Staff should perform hand hygiene before and after every client interaction, following CDC guidelines. Sharps containers must be puncture-resistant, clearly labeled, and replaced before reaching the fill line. For any business administering IV therapy, documented infection control policies reviewed annually help demonstrate compliance during regulatory audits.
How Should You Handle Biomedical Waste Disposal in Arizona?
You should handle biomedical waste disposal in Arizona by following the Arizona Department of Environmental Quality (ADEQ) regulations for biohazardous waste. IV therapy businesses generate regulated medical waste, including used needles, contaminated tubing, blood-soaked gauze, and empty IV bags with residual fluids.
Key disposal requirements include:
- Segregating sharps into FDA-cleared, puncture-resistant containers immediately after use.
- Placing biohazardous soft waste in red bags labeled with the universal biohazard symbol.
- Contracting with a licensed biomedical waste transporter for routine pickup and documented destruction.
- Maintaining waste tracking manifests for a minimum of three years.
Improper disposal can result in ADEQ citations and significant fines, making a contracted waste management partnership a practical necessity.
What Emergency Supplies and Medications Must Be on Hand?
The emergency supplies and medications that must be on hand include anaphylaxis response equipment, basic life support tools, and adverse reaction medications. Because IV therapy introduces substances directly into the bloodstream, allergic and anaphylactic reactions can occur without warning.
Essential emergency supplies include:
- Epinephrine auto-injectors for anaphylaxis.
- Diphenhydramine (Benadryl) for mild allergic reactions.
- Supplemental oxygen with a delivery mask.
- A bag-valve mask for respiratory support.
- Blood pressure cuffs, pulse oximeters, and stethoscopes for vital sign monitoring.
- A current, stocked crash cart or emergency kit appropriate to the clinical setting.
All clinical staff should maintain current Basic Life Support (BLS) certification at minimum. Routine checks ensuring medications are not expired and equipment is functional round out a compliant emergency preparedness program. With emergency protocols established, marketing those services responsibly requires its own set of compliance rules.
What Are the Requirements for Running a Mobile IV Therapy Business?
The requirements for running a mobile IV therapy business include proper licensing, compliant vehicle and equipment setups, and documented on-site client consent procedures. Each requirement is covered below.
Do Mobile IV Services Need a Separate License in Arizona?
Mobile IV services need the same foundational licenses as stationary IV therapy clinics, plus additional considerations based on where services are delivered. Arizona does not issue a single “mobile IV license.” Instead, operators must hold a valid Transaction Privilege Tax license, maintain medical director oversight, and ensure all nursing staff carry active Arizona Board of Nursing credentials.
Where it gets nuanced is at the city level. According to the City of Phoenix, no general business license is issued; instead, specific regulatory permits govern individual business activities. This means mobile operators serving clients across multiple municipalities should verify local permit requirements in each city. Failing to confirm city-level regulations before launching is one of the most common compliance oversights in the mobile IV space.
What Vehicle and Equipment Standards Apply to Mobile IV Therapy?
Vehicle and equipment standards for mobile IV therapy center on safe transport, proper storage, and clinical readiness at the client’s location. Vehicles must maintain temperature-controlled environments for IV fluids and medications, carry sharps disposal containers, and stock emergency supplies including epinephrine and oxygen.
According to a 2024 Grand View Research report, the U.S. mobile IV hydration therapy market was estimated at $568.5 million and is projected to grow at a CAGR of 10.4% through 2030. This rapid growth makes equipment standardization increasingly important. Basic startup costs for a mobile IV therapy setup typically range from $6,000 to $45,000, covering medical supplies, portable infusion equipment, and vehicle outfitting. Investing in quality portable equipment from the outset reduces both liability exposure and long-term replacement costs.
What Client Consent and Intake Procedures Are Required On-Site?
Client consent and intake procedures required on-site include documented informed consent, a health history questionnaire, allergy screening, and verification of a valid individualized medical order before infusion begins. Arizona Administrative Code requires healthcare providers to obtain informed consent from a client before providing treatment within the context of a professional relationship.
On-site intake should capture current medications, known allergies, and relevant medical conditions. Each consent form must clearly explain the specific IV treatment being administered, potential risks, and expected outcomes. Completing this documentation before any needle is inserted protects both the client and the provider.
With mobile operations properly structured, understanding total startup costs helps entrepreneurs plan their investment realistically.
How Much Does It Cost to Start an IV Hydration Business in Arizona?
The cost to start an IV hydration business in Arizona ranges from roughly $6,000 for a mobile setup to over $40,000 for a stationary clinic. Below, the key startup licensing fees and ongoing operational expenses are broken down.
What Are the Startup Licensing and Registration Costs?
The startup licensing and registration costs for an IV hydration business in Arizona include several state and local fees. Filing Articles of Organization for an LLC costs $50 for standard processing or $85 for expedited processing through the Arizona Corporation Commission. Same-day expedited service costs $200, requiring documents by 10:00 a.m. for processing by 5:00 p.m. The state Transaction Privilege Tax license adds $12 per location annually, while some cities charge additional fees; Scottsdale, for example, requires an additional $50 for its local TPT license. Professional liability insurance averages $91 per month for small businesses in Arizona, according to Insureon. When combined with initial inventory costs of $3,000 to $8,000 for IV supplies, bags, and kits, new owners should budget carefully before launch.
What Are the Ongoing Operational and Supply Costs?
The ongoing operational and supply costs for an IV hydration business include medical director fees, insurance premiums, inventory replenishment, and equipment maintenance. Medical director fees typically range from $1,000 to $3,000 per month depending on the level of involvement. Insurance remains a recurring expense, with professional liability averaging around $91 monthly. Restocking IV supplies, fluids, and vitamin additives represents one of the largest variable costs, since client volume directly drives consumption rates. According to Financial Models Lab, Initial medical equipment costs for a stationary clinic can reach approximately $40,000, and maintaining equipment adds ongoing expenses for any operation. For mobile operations, vehicle costs and fuel further increase the monthly overhead. Budgeting for these recurring expenses is just as critical as covering startup costs, since underestimating operational spend is one of the most common reasons new IV hydration businesses struggle in their first year.
With costs mapped out, understanding Arizona’s marketing and advertising rules helps protect that investment.
What Marketing and Advertising Rules Apply to IV Therapy in Arizona?
Marketing and advertising rules for IV therapy in Arizona fall under federal FTC and FDA guidelines for health claims and HIPAA regulations for patient privacy. The sections below cover prohibited claims and patient data protections.
What Health Claims Are Prohibited Under FTC and FDA Guidelines?
The health claims prohibited under FTC and FDA guidelines include any advertising statements that are untruthful, misleading, or unsupported by competent scientific evidence. According to the FTC’s health products compliance guidance, all health-related advertising claims must be truthful, not misleading, and backed by competent and reliable scientific evidence.
For IV hydration businesses, this means avoiding language that promises cures, guarantees specific outcomes, or overstates the therapeutic benefits of vitamin infusions. Claims like “IV therapy cures migraines” or “drip therapy eliminates chronic fatigue” would violate these standards without robust clinical data to support them.
Compliant marketing focuses on general wellness language, such as supporting hydration, promoting nutrient absorption, or complementing overall well-being. Every claim in advertising copy, social media posts, and website content should be reviewable against existing evidence before publication.
What HIPAA Considerations Apply to Marketing IV Services?
The HIPAA considerations that apply to marketing IV services center on protecting patient health information in all promotional activities. HIPAA marketing rules generally prohibit covered entities from using or disclosing Protected Health Information for marketing without valid written patient authorization.
Patient testimonials and before-and-after stories require particular caution. According to invigoMEDIA, digital marketing for healthcare services requires the de-identification of patient stories or case studies by removing 18 specific identifiers to remain HIPAA compliant. These identifiers include names, dates, geographic data, phone numbers, email addresses, and Social Security numbers.
Practical safeguards for IV therapy marketing include:
- Obtaining signed HIPAA-compliant authorization before using any patient information in advertising.
- Removing all 18 identifiers from testimonials, case studies, and social media content.
- Training staff on what constitutes Protected Health Information in digital marketing contexts.
- Reviewing email marketing lists to confirm patient data was not sourced from clinical records without consent.
For operators building a compliant IV hydration brand, these advertising and privacy frameworks form the foundation of trustworthy patient communication.
How Can a Nurse-Administered Mobile IV Service Meet These Requirements?
A nurse-administered mobile IV service can meet these requirements by integrating medical oversight, licensed clinical staff, and compliant protocols into a single on-demand care model. The following subsections cover how The Drip IV Infusion’s approach simplifies compliance and the key takeaways for Arizona IV hydration businesses.
Can The Drip IV Infusion’s Care Model Simplify Compliance?
Yes, The Drip IV Infusion’s care model can simplify compliance by combining nurse-led mobile delivery with built-in regulatory safeguards. Founded in Fall 2022 by professionals with decades of experience in nursing, emergency medicine, home health, and IV medical techniques, The Drip IV Infusion sends licensed nurses directly to clients’ homes or workplaces across Arizona.
This structure addresses several core requirements simultaneously:
- Every infusion is performed by a licensed nurse operating within the Arizona Board of Nursing’s scope of practice.
- Individualized medical orders guide each treatment, satisfying the state’s mandate for patient-specific protocols.
- Informed consent and health intake procedures are completed on-site before any IV is started.
- Proper documentation and record-keeping practices support the six-year medical records retention required under Arizona law.
Because The Drip IV Infusion operates as a mobile service, overhead remains lower than brick-and-mortar clinics while maintaining the same clinical and regulatory standards. For anyone evaluating how to meet Arizona’s IV hydration business requirements without building out a full facility, this nurse-administered mobile model offers a practical, compliance-focused path.
What Are the Key Takeaways About IV Hydration Business Requirements in Arizona?
The key takeaways about IV hydration business requirements in Arizona center on five areas: legal formation, medical oversight, nursing compliance, safety protocols, and marketing restrictions.
- Register your business entity with the Arizona Corporation Commission and obtain all required state and local tax licenses.
- Secure a qualified medical director who holds an active, unrestricted MD or DO license and provides documented oversight of all clinical protocols.
- Ensure every IV treatment is administered by a licensed RN or LPN under an individualized medical order from a provider who has examined the patient.
- Maintain OSHA compliance, proper infection control, biomedical waste procedures, and USP compounding standards where applicable.
- Follow FTC advertising rules and HIPAA privacy requirements for all marketing and patient communications.
According to the Goldberg Law Office, structuring these compliance layers correctly from the start is what separates a legally defensible IV hydration business from one that risks enforcement action. Arizona does not limit who can own an IV hydration business, but the clinical delivery must always flow through licensed professionals under physician oversight. Starting with a clear compliance framework protects the business and, most importantly, every client who receives care.